Research Work
Introduction:
Not only in India but worldwide, due to change in life style. Diabetes has become a very common hazardous disorder. It has hampered the creativity, intelligence and the active lifestyle of mankind; Most of researchers all over the world are actively involved in this antidiabetic study. Though we are stepping in the 21st century a modern era, the cause of diabetes & its curative therapy is still unknown. The conventional allopathic treatment has certainly its drawbacks. It has its side effects, drug intolerance, drug resistance, day to day increasing drug dosages & addition of newer drugs & finally shifting diabetic patient to injectable insulin. Patient has to face severe fluctuating BSL & resulting hazardous hypoglycaemia & coma. Allopathic therapy do not overcome diabetic complications like vasculopathy, neuropathy, nephropathy, retinopathy, cellulitis, memory loss, Irritability, sexual Impotence & so on.
After studying Allopathic, Ayurvedic, Unani, Homeopathic medication we concluded that there is no appropriate medication to cure diabetes & its complications. India being a tropical country, has varied climate. Various medicinal plants are found here & we took ben/fit of this herbal wealth. We scanned hundreds of plants & after a deep study we formulated a complete, excellent, herbal medicine "Mehanorm"
AIMS AND OBJECTIVES:
The present study of the clinical trials on a new herbal antidiabetic formulation has been carried out with the following objectives
EXPERIMENTAL WORK:
Literature review from various systems of medicine indicated the great potential of herbs drugs as antidiabetic agents. The crude drugs were selected, collected and authenticated from reliable sources. According to their use, crude drugs were categorised into different categories like Hypoglycaemia, Neuropathy, Retinopathy, Nephropathy, Vasculopathy, & even as an immunomodulators. Finally by considering easy availability, cost, potency, compatibility, we selected few crude drugs. They were dried, powdered & sieved ( size no. 22) mixed in appropriate proportion and 5 gm. of material of above drug formulation is boiled with 400m1of water and concentrated to 100m1. This 100 ml decoction (Mehanorm) is used in the patient as a single dose, orally empty stomach in morning hours.
PRE-CLINICAL STUDIES:
Toxicity Study: Acute toxicity studies were carried out in 30 Swiss albino mice divided in five groups. Group 1 to 4 was given formulation in varying doses while the 5th group was kept the as control group Mehanorm was found to be safe up to a high dose of 2 gm./kg/day without any toxic reaction.
ANTIDIABETIC ACTIVITY STUDY:
For the antidiabetic activity Alloxan induced diabetes model was used. 18 rabbits were divided into three groups - as control, standard and test groups. Chlorpropamide was taken as a standard. Test group was treated for 8 days with Alloxan monohydrate (60 mg/kg) to induce diabetes. The blood sugar level and urine samples were checked daily throughout the experiment. The results of ant/diabetic activity are given in Table below.
| Group | BSL + S.D. | |||||
| Intial | 0.5 hrs | 1 | 2 | 3 | 4 | |
| Diabetic Control | 230 | 232 | 235 | 238 | 242 | 244 |
| 7.98 | 7.70 | 7.93 | 7.81 | 8.30 | 8.06 | |
| Drug Treated | 226 | 220 | 205 | 161 | 127 | 115 |
| 4.56 | 3.88 | 7.24 | 9.58 | 6.56 | 4.18 | |
| Standard | 232 | 225 | 215 | 185 | 162 | 135 |
| 5.08 | 6.34 | 8.21 | 3.59 | 4.22 | 5.50 | |
CLINICAL STUDIES:
We started our development of antidiabefic formulation in 1993 and clinical studies on human individuals from last 15years.
Mehanorm was prescribed in a daily dose of 5gm/per day in the form of decoction on empty stomach. According of disease the dose may be varied up to thrice daily. Treatment is given irrespective of age, sex, severity of disease, associated complications and concomitant medication. Following table of 30 patients represents our clinical studies for varying duration of Mehanorm therapy.
Blood Sugar level before and after the Mehanorm therapy
| Patients | (1Yr.) Long Duration | (6 mon.) Intermediate duration | (1 mon.)Short Duration | |||
| Before | After | Before | After | Before | After | |
| 1 | 227 | 148 | 154 | 137 | 201 | 140 |
| 2 | 286 | 151 | 190 | 160 | 198 | 160 |
| 3 | 265 | 159 | 246 | 151 | 198 | 170 |
| 4 | 219 | 148 | 251 | 155 | 182 | 163 |
| 5 | 296 | 125 | 207 | 145 | 416 | 251 |
| 6 | 248 | 131 | 182 | 86 | 224 | 150 |
| 7 | 236 | 145 | 212 | 154 | 255 | 140 |
| 8 | 241 | 170 | 258 | 192 | 257 | 108 |
| 9 | 305 | 186 | -- | -- | 238 | 168 |
| 10 | 198 | 101 | -- | -- | 248 | 131 |
CLINICAL FINDINGS OF Mehanorm THERAPY
Mehanorm dose was adjusted considering the BSL and severity of disease without changing the patient’s concurrent anti-diabetic treatment. During follow up by considering the BSL we tapered the dose of allopathic medication, without altering Mehanorm dose. Gradually then allopathic medication was stopped within 3 to 6 months as per BSL to avoid hypoglycemia. Then Mehanorm dose was tapered as per BSL gradually to once daily dose.
From this we inferred that with Mehanorm therapy the BSL was under excellent control and the patient was also relieved of the diabetic complications.
Here is the graphical representation of BSL of randomly selected 700 patients, amongst more than 10,000 patients on these patients were categorised into 4 groups as under:
Effect of Mehanorm on Fasting and Post Prandial Blood Sugar Values in Newly detected Patients (Group I)
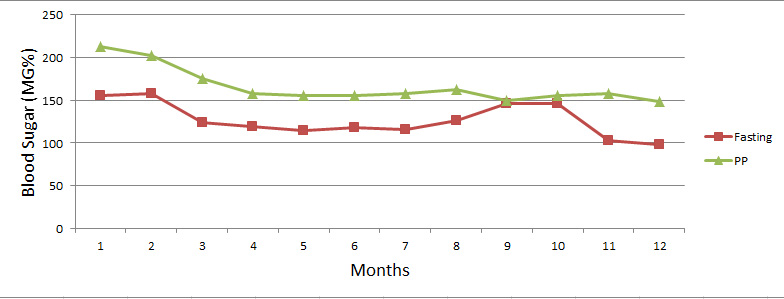
Group I: Graph shows significant effect of Mehanorm in newly detected patients. These patients were under excellent BSL NI control within 2 Yrs. of therapy. Approximately 20% of BSL was reduced within first 3 —4 months of Mehanorm therapy. )BSL was maintained for next 6 months after that concomitant allopathic medication was stopped This raised BSL in 8th —10th month of therapy. This raised BSL Mehanorm stimulates insulin secretion, which stabilizes BSL within normal range.
| Months | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Fasting | 156 | 158 | 124 | 119 | 115 | 118 | 116 | 126 | 146 | 146 | 103 | 98 |
| PP | 213 | 202 | 175 | 158 | 155 | 155 | 158 | 162 | 150 | 155 | 158 | 148 |
Effect of Mehanorm on Fasting and Post Prandial Blood Sugar Values in NIDDM Patients on OHA (Group II)
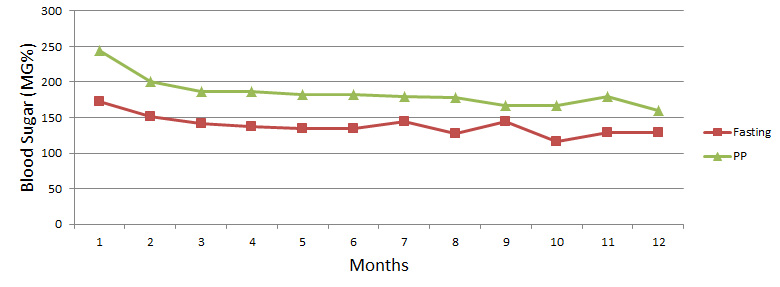
Group II: In-group II with Mehanorm therapy BSL reduced to 10% in 1st month. So OHA (Oral Hypoglycaemic Agent) dose was gradually reduced. Which shows some ups & downs in BSL up to 6 months. After that concomitant OHA was stopped, which raised BSL. This raised BSL & 6yursulin stimulates insulin secretion, so BSL dropped to fasting BSL 96 mg% & PP BSL 166 mg% in 16th month. Then BSL was maintained to normal range i.e. fasting BSL 113 mg% & PP BSL 138 mg% with only Mehanorm therapy.
With Mehanorm dose thrice a day & reducing OHA dose gradually within 1st 6 months, all diabetic symptoms and complications were relived up to 80%. Then OHA was stopped & Mehanorm dose was tapered to once daily. We hope in future patient will be completely cured of DM with Mehanorm therapy.
| Months | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Fasting | 172 | 152 | 141 | 137 | 135 | 135 | 145 | 127 | 144 | 117 | 129 | 129 |
| PP | 244 | 200 | 186 | 186 | 182 | 183 | 179 | 178 | 167 | 167 | 180 | 160 |
Effect of Mehanorm on Fasting and Post Prandial Blood Sugar Values in NIDDM Patients on Insulin (Group III)
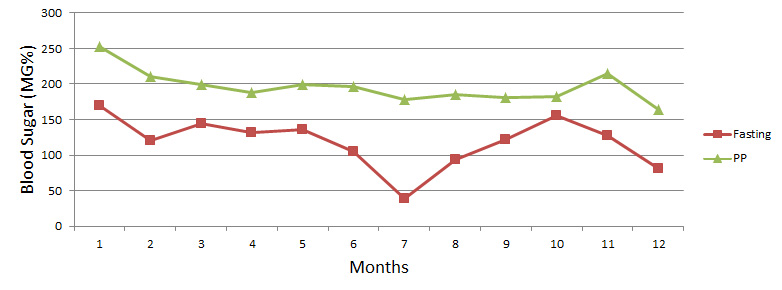
Group III: Graph III shows reduction of BSL by 25% within 1st month of Mehanorm therapy. There are some ups & downs in BSL as the dose of insulin was reduced gradually & then stopped completely between 10-12 months, keeping Mehanorm dose constant i.e. thrice a day. Patients were also relieved from all symptoms and diabetic complications.
The observation during therapy suggests that Mehanorm regenerates 13-cells of Langerhans & improves peripheral insulin sensitivity. So we found that Mehanorm has additive effect along with insulin.
| Months | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Fasting | 170 | 120 | 145 | 132 | 136 | 105 | 39 | 94 | 122 | 155 | 128 | 81 |
| PP | 253 | 211 | 199 | 188 | 199 | 196 | 178 | 185 | 181 | 183 | 215 | 164 |
Effect of Mehanorm on Fasting and Post Prandial Blood Sugar Values in IDDM Patients (Group IV)

Group IV: Graph shows reduction of BSL by 45% within 1st month of Mehanorm therapy, keeping the Insulin dose constant. This indicates that Mehanorm improves the peripheral insulin sensitivity.
By keeping Mehanorm dose constant; insulin dose was gradually reduced & then even stopped in few patients. This shows Aygu4in regenerates B-Cells of Langerhans.
Patients were also relieved from nearly all symptoms and Diabetic complications.
This observation indicates that though. Mehanorm herbal oral hypoglycaemic agent, it has good scope in IDDM patients
| Months | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Fasting | 171 | 97 | 128 | 154 | 133 | 80 | 80 | 80 | 86 | 86 | 86 | 86 |
| PP | 272 | 144 | 206 | 234 | 230 | 233 | 195 | 195 | 142 | 220 | 220 | 220 |
Conclusion:
Second Preclinical Study conducted at Grant Medical College, Mumbai.
STUDY REPORT:
Efficacy and safety of "Mehanorm” [A Polyherbal formulation] in Alloxan induced Diabetes Mellitus in Albino Rats.
STUDY CENTRE
Department of Pharmacology. Grant Medical Collage.
Byculla, Mumbai - 400 008.
STUDY DURATION
Data Collection 8 Weeks
Data analysis and report preparation 2 Weeks
Total duration 10 Weeks
RESULT & CONCLUSION
Table 1 shows the demographic data and the baseline values for fasting blood sugar of the animals used in the study. All the three groups were compared with respect to the mean age, number of animals, mean fasting blood sugar before Alloxan and in the diabetic state (7 days after Alloxan).
Table 1: Demogratic data baseline parameters at enrolment in the groups.
| Parameter | AYURSULIN (n=6) |
Glibenclamide (n=6) |
Gum Acacia (n=6) |
ANOVA | |||
| No. of Animals | 6 | 6 | 6 | ||||
| Weight (Grams) | 136.67 | 8.16 | 138.33 | 7.53 | 137.50 | 9.87 | p>0.05 [N.S.] |
| Age (Days) | 69.83 | 7.52 | 72.83 | 4.99 | 70.17 | 6.94 | p>0.05 [N.S.] |
| Fasting Blood Sugar Before Alloxan (mg/dl) | 74.67 | 12.56 | 70.33 | 10.59 | 72.83 | 14.66 | p>0.05 [N.S.] |
| Fasting Blood Sugar 7 Days after Alloxan (mg/dl) | 122.17 | 9.57 | 118.17 | 13.55 | 118.67 | 14.33 | p>0.05 [N.S.] |
Table 2 & figure 1 shows the mean fasting blood sugar before Alloxan, in the diabetic state (7 days after Alloxan) and at weekly intervals up to 6 weeks during the study period. It is evident that there is significant reduction in the fasting blood sugar with Mehanorm (From 122.17 to 76.33 7.67 after 6 weeks) and Glibenclamide (From 118.17 13.55 to 78.33 7.33 after 6 weeks beginning from the first week of study medication and this reduction was statistically significant (p<0.01, Repeat measures ANOVA) during the entire 6 weeks of study period. The reduction in blood sugar is greater with Mehanorm as compared to glibenclamide (Figures 2 & 3). There is no statistically significant reduction (p>0.05, Repeat measured ANOVA) in the fasting blood sugar in the gum acacia (Vehicle) group (118.67 14.33 to 106.67 9.67).
Table 2: Fasting Blood Sugar (Mean S.D.) in the study groups in normal, diabetic and after weekly intervals up to 6 weeks of study treatment.
| Parameter | AYURSULIN (n=6) |
Glibenclamide (n=6) |
Gum Acacia ANOVA (n=6) |
| Before Alloxan | 74.67 12.56 | 70.33 10.59 | 72.83 14.66 p>0.05 [N.S.] |
| Diabetic Rats (7 days after Alloxan) |
122.17 9.57 | 118.17 13.55 | 118.67 14.33 p>0.05 [N.S.] |
| Week 1 | 100.15 7.91 | 96.25 10.67 | 114.58 8.94 p=0.049 [Sig.] |
| Week 2 | 91.50 7.85 | 95.83 9.67 | 108.17 9.33 p=0.032 [Sig.] |
| Week 3 | 84.66 8.33 | 89.51 9.33 | 105.29 8.67 p=0.0078 [Sig.] |
| Week 4 | 74.33 7.67 | 84.33 9.67 | 104.67 9.33 p=0.056 [Sig.] |
| Week 5 | 76.32 8.33 | 79.82 8.33 | 104.28 8.67 p=0.0029 [Sig.] |
| Week 6 | 76.33 7.67 | 78.33 7.33 | 106.67 9.67 p=0.0007 [Sig.] |
| Repeat measures | p=0.0014 | p=0.0049 | p>0.05 |
| ANOVA [After Alloxan] |
[Sig.] | [Sig.] | [N.S.] |
Table 3 & figure 4 show the weight of the animals before and after 6 weeks of study period. There is no significant change in the body weight in Mehanorm and Glibenclamide groups whereas in the gum acacia group there is a significant increase in the body weight seen (p<0.05, paired 't'-test).
Table 3 : Weight (Mean S.D.) of the animals in the study groups at enrolment and after 6 weeks of the study period.
| Parameter | AYURSULIN (n=6) |
Glibenclamide (n=6) |
Gum Acacia (n=6) |
ANOVA |
| At Baseline (7 Days after Alloxan |
136.67 8.16 | 138.33 7.53 | 137.50 9.87 | p>0.05 [N.S.] |
| After 6 Weeks | 144.67 7.33 | 147.33 12.67 | 186.67 12.67 | p>0.05 [N.S.] |
| Paired 't' - test | p>0.05 [N.S.] |
p>0.05 [N.S.] |
p<0.05 [Sig.] |
Table 4 shows the laboratory parameters and gross weight of liver & kidneys in the three treatment groups before and at the end of 6 weeks of study period. There is no significant change in the mean values for bilirubin, alkaline phosphatase, SGPT, blood urea nitrogen, creatinine and gross weight of liver and kidneys in the three treatment groups after 6 weeks of study period (p>0.05, One-way ANOVA). Also there is no significant difference between the normal animals and the study groups for all the above values.
Table 4 : Laboratory parameters (Mean S.D.) in the normal animals & study groups after the completion of study period of 6 weeks.
| Parameter | Normal (n=6) |
After 6 weels of Study Period | |||||||
| AYURSULIN (n=6) |
Glibenclamide (n=6) |
Gum Acacia (n=6) |
ANOVA | ||||||
| Bilirubin | 1.24 | 0.33 | 1.34 | 0.67 | 1.36 | 0.33 | 1.67 | 0.67 | p>0.05 [N.S.] |
| Alkaline Phosphates | 18.42 | 5.67 | 19.38 | 6.33 | 18.56 | 7.33 | 1.36 | 8.67 | p>0.05 [N.S.] |
| SGPT | 16.98 | 5.33 | 16.32 | 5.67 | 17.53 | 6.67 | 18.16 | 7.33 | p>0.05 [N.S] |
| BUN | 20.72 | 7.67 | 22.68 | 8.33 | 23.81 | 9.33 | 23.34 | 10.33 | p>0.05 [N.S.] |
| Sr. Creatinine | 1.24 | 0.33 | 1.34 | 0.67 | 1.36 | 0.33 | 1.67 | 0.67 | p>0.05 [N.S.] |
| Gross Weight (gms) | |||||||||
| Liver | 7.67 | 1.33 | 7.33 | 1.33 | 5.67 | 0.67 | 7.67 | 1.33 | p>0.05 [N.S.] |
| Kidneys (both) | 6.37 | 0.67 | 6.67 | 1.33 | 5.67 | 1.33 | 6.67 | 0.67 | p>0.05 [N.S.] |
On histopathological examination of liver and kidneys, no charged were seen in the study groups after 6 weeks of the study period.
Thus, Mehanorm has shown a significant improvement in the fasting blood sugar levels Alloxan induced diabetes in male albino ratsthis reduction is similar to glibenclamide and superior to placebo (vehicle).
